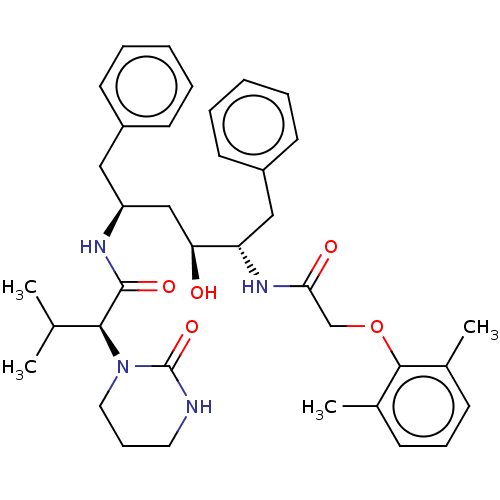
 med.21724, Compound 185 ABT-378 A-157378-0 BDBM50180655 Lopinavir CHEBI:31781 A-157378.0 Kaletra
med.21724, Compound 185 ABT-378 A-157378-0 BDBM50180655 Lopinavir CHEBI:31781 A-157378.0 Kaletra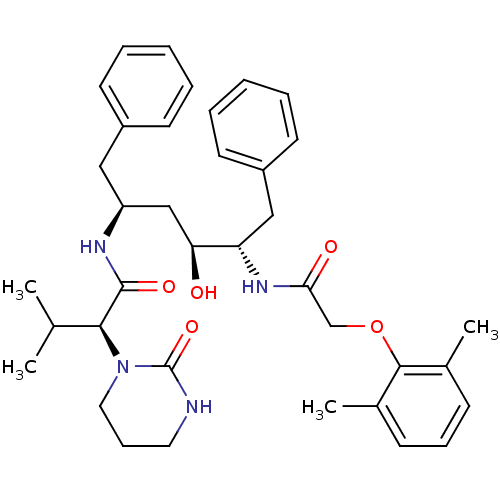 CHEMBL729 ABT-378 BDBM578 LPV Aluviran Lopinavir (2S)-N-[(2S,4S,5S)-5-[2-(2,6-dimethylphenoxy)acetamido]-4-hydroxy-1,6-diphenylhexan-2-yl]-3-methyl-2-(2-oxo-1,3-diazinan-1-yl)butanamide
CHEMBL729 ABT-378 BDBM578 LPV Aluviran Lopinavir (2S)-N-[(2S,4S,5S)-5-[2-(2,6-dimethylphenoxy)acetamido]-4-hydroxy-1,6-diphenylhexan-2-yl]-3-methyl-2-(2-oxo-1,3-diazinan-1-yl)butanamide
- Vishnuvardhan, D; Moltke, LL; Richert, C; Greenblatt, DJ Lopinavir: acute exposure inhibits P-glycoprotein; extended exposure induces P-glycoprotein. AIDS 17: 1092-4 (2003)
- Sham, HL; Betebenner, DA; Herrin, T; Kumar, G; Saldivar, A; Vasavanonda, S; Molla, A; Kempf, DJ; Plattner, JJ; Norbeck, DW Synthesis and antiviral activities of the major metabolites of the HIV protease inhibitor ABT-378 (Lopinavir). Bioorg Med Chem Lett 11: 1351-3 (2001)
- Hidaka, K; Kimura, T; Sankaranarayanan, R; Wang, J; McDaniel, KF; Kempf, DJ; Kameoka, M; Adachi, M; Kuroki, R; Nguyen, JT; Hayashi, Y; Kiso, Y Identification of Highly Potent Human Immunodeficiency Virus Type-1 Protease Inhibitors against Lopinavir and Darunavir Resistant Viruses from Allophenylnorstatine-Based Peptidomimetics with P2 Tetrahydrofuranylglycine. J Med Chem 61: 5138-5153 (2018)

